damian.bond@pharmakure.com
damian.bond@pharmakure.com
PharmaKure Announces Successful Scientific Advice Meeting with the MHRA.
Manchester, United Kingdom 6th February 2023
PharmaKure Sponsors the Multiple Systems Atrophy (MSA) Research Symposium at UCL on the 20th January, 2023.
Manchester, United Kingdom 20th January 2023
PharmaKure Announces Move of Headquarters to Circular 1 Health in Didsbury Manchester
Manchester, United Kingdom 17th October 2022
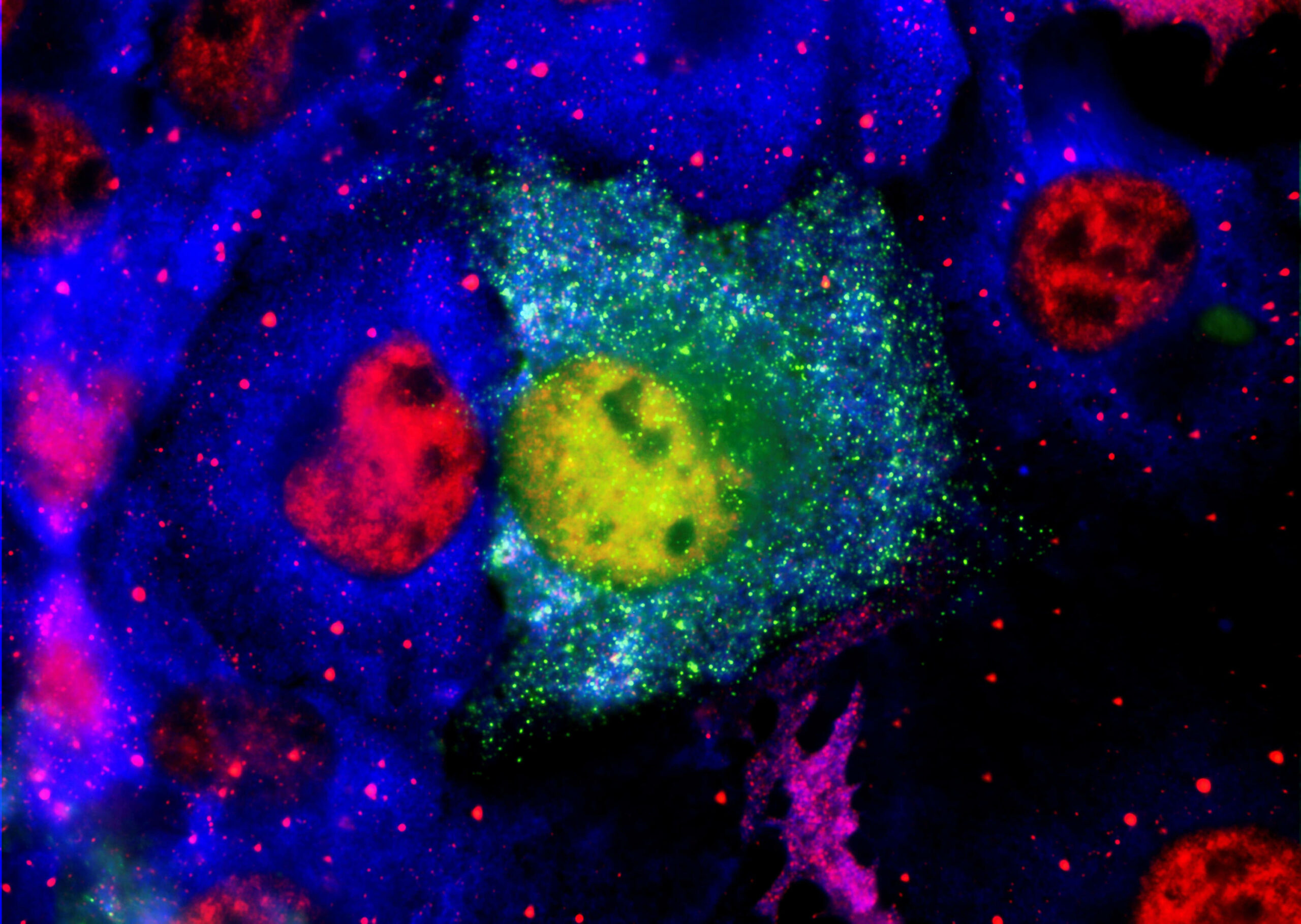
PharmaKure has launched a study to identify blood-based biomarkers in patients with Alzheimer’s Disease.
Manchester, United Kingdom 4th October 2022
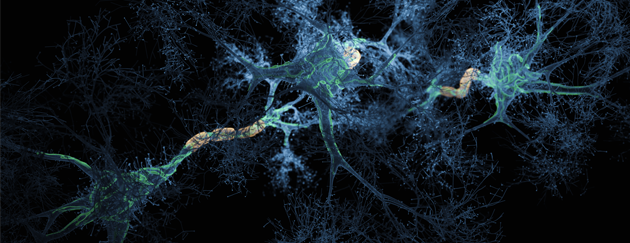
The latest news on Alzheimer’s disease
Links to sites providing the lates updates on research, news, help and support for Alzheimer's disease.
The latest news on Dementia with Lewy Bodies
Links to sites providing the lates updates on research, news, help and support for Dementia with Lewy Bodies.
The latest news on Gerstmann-Straussler-Scheinker disease
Links to sites providing the lates updates on research, news, help and support for Gerstmann-Straussler-Scheinker disease.
Drug Repurposing: progress, Challenges and recommendations
Abstract Given the high attrition rates, substantial costs and slow pace of new drug discovery and development, repurposing of ‘old’ drugs to treat both common and rare diseases is increasingly becoming an attractive proposition because it involves the use of de‑risked compounds, with potentially lower overall development costs and shorter development timelines. Various data-driven and experimental approaches have been suggested for the identification of repurposable drug candidates; however, there are also major technological and regulatory challenges that need to be addressed. In this Review, we present approaches used for drug repurposing (also known as drug repositioning), discuss the challenges faced by the repurposing community and recommend innovative ways by which these challenges could be addressed to help realize the full potential of drug repurposing.
Differential Expression of AMPA Subunits Induced by NMDA Intrahippocampal Injection in Rats
Abstract Glutamate is involved in excitotoxic mechanisms by interacting with different receptors. Such interactions result in neuronal death associated with several neurodegenerative disorders of the central nervous system (CNS). The aim of this work was to study the time course of changes in the expression of GluR1 and GluR2 subunits of glutamate amino-acid-3-hydroxy-5-methyl-isoxazol-4-propionic acid (AMPA) receptors in rat hippocampus induced by NMDA intrahippocampal injection. Rats were submitted to stereotaxic surgery for NMDA or saline (control) microinjection into dorsal hippocampus and the parameters were evaluated 24 h, 1, 2, and 4 weeks after injection. The extension and efficacy of the NMDA-induced injury were evaluated by Morris water maze (MWM) behavioral test and Nissl staining. The expression of GluR1 and GluR2 receptors, glial fibrillary acidic protein (GFAP), and neuronal marker (NeuN) was analyzed by immunohistochemistry. It was observed the impairment of learning and memory functions, loss of neuronal cells, and glial proliferation in CA1 area of NMDA compared with control groups, confirming the injury efficacy. In addition, NMDA injection induced distinct changes in GluR1 and GluR2 expression over the time. In conclusion, such changes may be related to the complex mechanism triggered in response to NMDA injection resulting in a local injury and in the activation of neuronal plasticity.
Inhibition of protein aggregation and amyloid formation by small molecules
Abstract For decades, drug after drug has failed to slow the progression of Alzheimer’s disease in human trials. How compounds reducing fibril formation in vitro and toxicity in transgenic mice and flies bind to the Ab toxic oligomers, is unknown. This account reviews recent drugs mainly targeting Ab, how they were identified and report their successes from in vitro and in vivo experimental studies and their current status in clinical trials. We then focus on recent in vitro and simulation results on how inhibitors interact with Ab monomers and oligomers, highly desirable knowledge for predicting new efficient drugs. We conclude with a perspective on the future of the inhibition of amyloid formation by small molecules